Today, Lennox Industries announced its PureAir™ and PureAir™ S Air Purification Systems, with the Lennox Healthy Climate™ Carbon Clean 16® air filter, remove over 99% of the virus that causes COVID-191 from the air2. After months of rigorous testing at a highly reputed, independent laboratory, this announcement is further evidence of Lennox’ relentless commitment to delivering unmatched, whole-home air purification for perfect air.
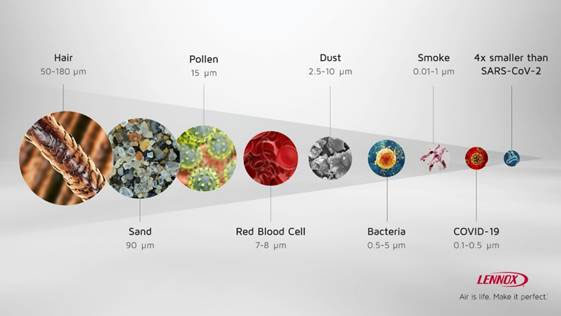
Lennox’ residential air filtration systems are hospital-grade3 air filtration systems that remove over 99% of virus particles, including the common cold, influenza and viruses 4x smaller than SARS-CoV-2. They are even more efficient – 99.9% to be precise – at removing larger particles, like mold spores, pollen, dust and pet dander. Unlike other air purification systems, the PureAir™ and PureAir™ S systems produce no ozone, a common lung irritant. Better still, they actively work to reduce ozone generated by other sources in your home.

“This year, more than ever, we all recognize the critical importance of clean air and how it is vital to our health,” said Quan Nguyen, Vice President and General Manager, Lennox Industries Residential. “At Lennox, we have long pursued the highest level of indoor air quality on behalf of our customers. During this pandemic, we’ve increased our investments in air purification technologies in our pursuit of perfect air, so consumers can breathe easy.”
The Lennox Healthy Climate™ Carbon Clean 16® air filter was tested by LMS Technologies, Inc., an independent filtration research and testing lab that has been used by Lennox for over two decades. The lab performs unbiased, controlled testing with standardized test procedures to ensure statistically verified results.
When paired with the PureAir™ S Air Purification System or the Lennox Healthy Climate™ Carbon Clean 16® air filter, a Lennox HVAC system provides unrivaled air filtration and the ultimate level of indoor air quality to ensure the health and safety of homeowners and their families. Part of Lennox’ Ultimate Comfort System™, the PureAir™ S air purifier offers seamless precision in air quality and intelligently works with other units in the Ultimate Comfort System™ to stay finely tuned to the home for reliably clean air.
“We understand that indoor air quality is a higher priority now more than ever before,” said John Whinery, VP of Product, Lennox Industries. “While COVID-19 has changed the world around us, it hasn’t changed our unwavering commitment to delivering consistently clean, comfortable air to homeowners through our Lennox Healthy Climate™ and Ultimate Comfort System™ indoor air quality products.”
For more information on Lennox’ product capabilities and ongoing dedication to the health and wellness of consumers, please visit Lennox.com/Residential.
About Lennox Industries
Lennox Industries, a division of Lennox International Inc. (NYSE: LII), is in constant pursuit of perfect air because we believe everyone deserves it. Whatever your version of perfect is, we can help make it happen. Offering some of the quietest and most efficient units on the market, the first ultra-low emissions furnace, and hospital-grade air filtration, Lennox has a history of designing innovative heating, cooling and indoor air quality products. Lennox’ approach to product design has earned it more Dealer Design awards than its competitors combined and multiple ENERGY STAR Most Efficient certifications. Lennox is a proud contributor to its communities, investing in the future of the HVAC industry and giving back through its corporate social responsibility program, Feel The Love. For more information about how Lennox is redefining air, visit Lennox.com/Residential.
- When used properly with other best practices recommended by CDC and others, filtration can be part of a plan to reduce the potential for airborne transmission of COVID-19 indoors.
- Removal efficiency based on third party testing results using MS-2 bacteriophage (ATCC 15597-B1). Bacteria representative of virus-sized particles like SARS-CoV-2, the virus that causes COVID-19. November 2020.
- Not intended for actual hospital use. Based on Table 1: Application guidelines in ANSI/ASHRAE Standard 52.2-2017.














